
Office: IFNH 250
Lab: IFNH 220
Phone: 848-932-0266
Email: harini.sampath@rutgers.edu
The overarching goal of the Sampath lab is to delineate the cellular alterations that occur during the development of metabolic diseases, including fatty liver, cardiovascular disease, and diabetes. In this way, we seek to identify pathways that can be targeted for nutritional and pharmaceutical therapies to preserve health and improve health outcomes. Current projects in the lab can be grouped into the following topics:
Regulation of lipid metabolism and its impact on tissue and whole body health
This project has direct relevance to understanding human diseases like atherosclerosis, fatty liver, and diabetes and devising nutritional and pharmacological approaches to mitigate these metabolic diseases. Specifically, we are interested in understanding how lipid metabolism is regulated in different cell types and tissues like liver, intestine, and heart, with an emphasis on the lipid modifying enzyme, stearoyl-CoA desaturase (SCD). SCD catalyzes the conversion of saturated fatty acids to monounsaturated fatty acids. SCD is a highly regulated enzyme, despite the fact that its products are abundant in our diets. In turn, it plays a critical role in regulating cellular metabolism: animals lacking SCD1 are lean and protected from metabolic disease; and in human cohorts, increased SCD activity is associated with metabolic syndrome. These prior findings have rendered SCD a very attractive target for manipulation of cellular metabolism. Current investigations in our lab are focused on delineating the tissue-specific roles for this highly regulated enzyme and identifying novel roles in understudied tissues. To do this, we use unique transgenic mouse models developed in our lab, as well as primary and immortalized cell lines in culture. We use a combination of cell biological, biochemical, and molecular biology tools to address our questions related to regulation of lipid metabolism and its role in overall health.
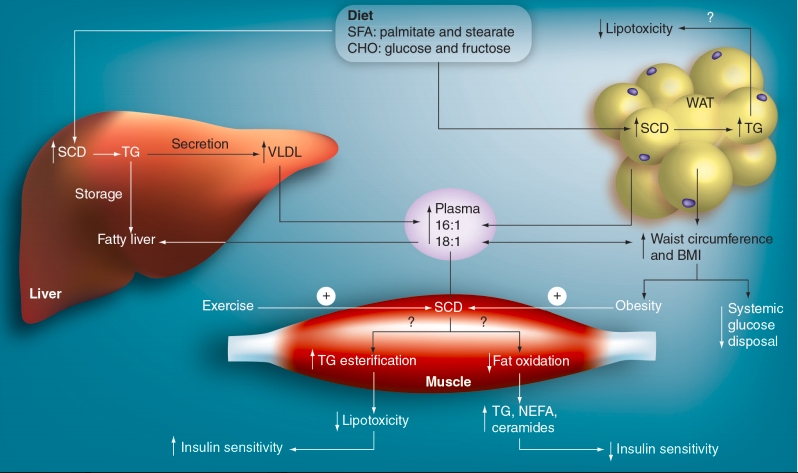
Figure credit: Adapted from Sampath and Ntambi. Future Lipidology 2008 3:163-73.
Oxidative DNA damage and repair and implications to muscle function
This project is focused on identifying novel mechanisms by which muscle function and endurance may be improved and devising strategies to preserve muscle function during the aging process. Oxidative stress, a normal cellular process that is exacerbated by aging or by consumption of sub-par diets, results in damage to DNA pools within the cell. Such oxidative DNA damage is repaired via the base excision repair (BER) pathway . We study how defects or enhancements in the BER pathway may be utilized to improve muscle function and endurance, via modulation of mitochondrial health. By using novel transgenic animals, isolated myotubes, and muscle cells in culture, a direct translational goal of these studies is to devise novel means to mitigate age-related losses in muscle function.
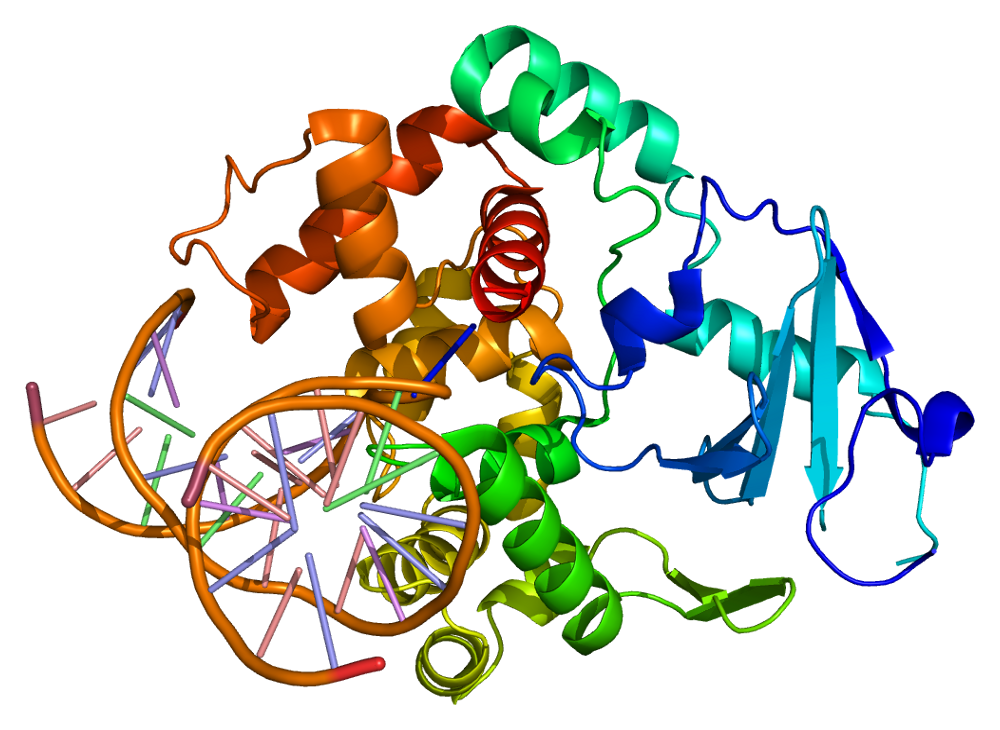
Figure credit: wikipedia.org
Influence of dietary flavonoids on the gut microbiome - sex differences and implications for metabolic health
This project explores sex differences in response to dietary flavonoids and modulation of the gut microbiome. A direct goal is to develop personalized nutrition strategies to preserve metabolic and gut health. By studying divergent responses to oral nutrition therapies in male and female animals and combining multiple 'omics' approaches (microbiomics, genomics, metabolomics, lipidomics), we aim to identify unique pathways that are regulated by dietary flavonoids and ultimately influence overall health.
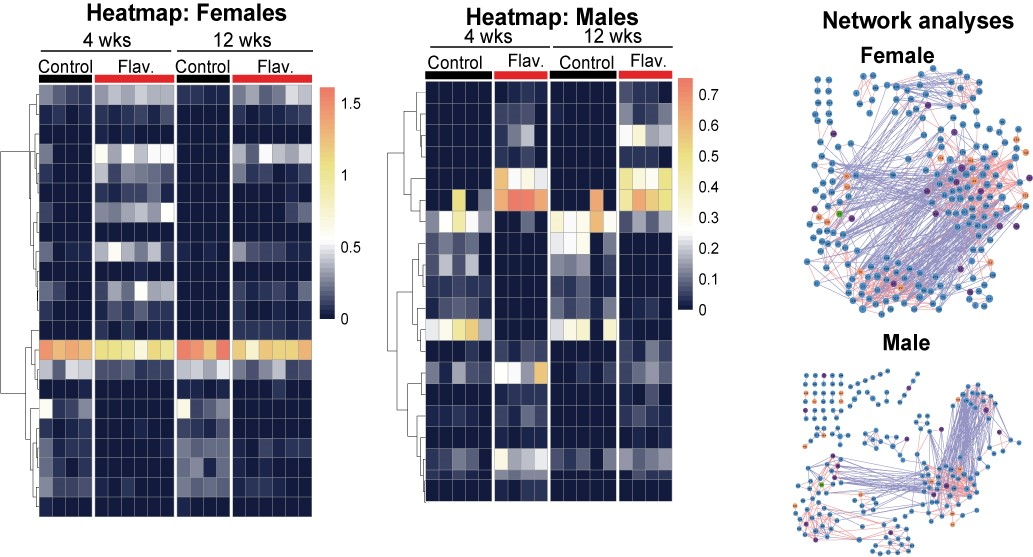
Figure credit: Sampath Lab
Join us: We are a group of energetic and diverse individuals with a passion for understanding how nutrients and genetics influence overall health. Alumni from the lab have continued their careers both in academia and in the private sector. The lab is housed within the brand-new Institute for Food, Nutrition, and Health (IFNH). We are always interested in welcoming talented postdoctoral fellows and graduate and undergraduate students to the team. For more information, please visit: https://sampathlab.com/.
Selected Publications
For a complete list of publications, please see here.
For Google Scholar, see: https://scholar.google.com/citations?hl=en&user=lIcrufMAAAAJ


