
Lipids such as fatty acids and cholesterol are involved in innumerable cellular processes, including energy storage and production, membrane biogenesis, signal transduction, and the regulation of gene expression. Nevertheless, the mechanisms by which lipids are transported and targeted within cells remain incompletely understood. Abnormal lipid trafficking, such as that occurring in lipid-storage diseases, can lead to severe cellular pathologies. The overall focus of research in this laboratory is on lipid traffic in cells, with particular emphasis on long-chain fatty acids, monoacylglycerols, and cholesterol. Ongoing efforts are addressing the following questions:
- What are the functions of cytoplasmic fatty acid-binding proteins (FABP) in intracellular lipid transport and metabolism? Why do different cell types have different FABP? How do the intestinal FABP’s impact whole body energy homeostasis?
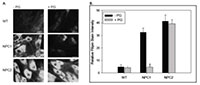
- How does the intracellular cholesterol-binding protein, Niemann-Pick type C2 protein (NPC2), function in the transport of cholesterol out of endosomes and lysosomes, and can we use specific phospholipid enrichment to treat NPC disease?
 Zoom in
Zoom in
The laboratory uses a combination of biochemical, biophysical, cell and molecular biological, and whole animal studies to answer these questions. For example, the structural determinants of the lipid transport properties of FABP and NPC2 proteins are probed using site-directed mutagenesis, fluorescence spectroscopy, and membrane biophysics approaches. Cell culture systems such as the Caco-2 human intestinal cell line are used to examine cellular lipid transport using biochemical methods and confocal microscopy. The functions of FABPs are also studied using transgenic mouse models in which specific FABP expression has been "knocked out". Cells from patients with NPC disease are used to examine the function of the NPC2 and NPC1 proteins in cholesterol trafficking. These studies are providing fundamental information about the cellular trafficking of lipids, with the ultimate goal of enabling effective preventive and therapeutic approaches to a variety of pathologic conditions including obesity, cardiovascular disease, and lipid-storage diseases.


